 
Thus, cesium- ion (Cs+) has eight valence electrons.Ĭs+ valency is not zero like noble gas as their outermost 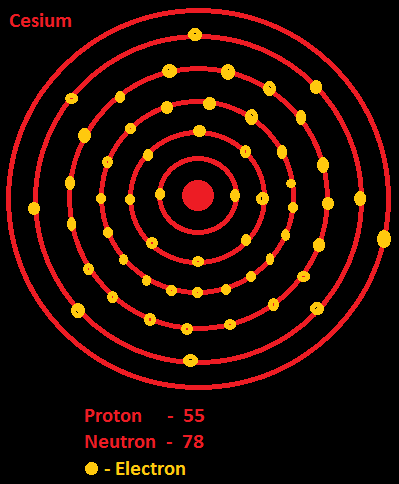
The electron configuration of neutral Cs is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 5s 2 5p 6 6s 1 but in Cs+ it loses one electron, so it has a new electron configuration of 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 5s 2 5p 6 means Cs+ has (2+6=8) outermost electrons which makes it stable. Valence electrons and valency of Cs+Ĭesium-ion Cs+ means it has lost one electron and has onlyġ0 electrons in the orbitals. Metals group and valency of alkali metals are always 1. as cesium is an element of group 1 which indicated alkali We can also find the valency of cesium with the help of a periodic table. Gas configuration) by losing one outermost electron. noble gases).Īlkali metals like cesium reached the stable (nearest inert Possess both positive and negative valency and atoms having eight outermostĮlectrons have zero valencies (i.e. Outermost shells is between one to four, the atom has positive valency and ifĮlectrons are between four to eight, the valency is calculated by subtractingįrom eight and valency is negative. Valency of an atom is determined based on the number of electrons lost, gained, or shared with another atom.Īn atom is said to be stable when its outermost shells haveĮight electrons (except H and He). 
How easily an atom or a free radical can combine with other chemical species. There are many different ways to find out the valency of anĪtom which reflects the ability of an atom to bond with other atoms. Of an atom is called valence electrons, and there is only one electron present The total number of electrons present in the valence shell Less) in any condition for a particular atom and may or not be equal to itsĪs we know, the valence shell of an atom can be found from the highest number of principle quantum numbers which is expressed in the term of n, and in 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 5s 2 5p 6 6s 1, the highest value of n is 6 so that the valence shell of Cs is 6 s 1. The valenceĮlectrons for a neutral atom is always definite, it cannot be varied (more or Valence electrons are the total number of electrons present You have to know what these two terms are, so without wasting your time let's goįor it, Difference between valence electrons and valency Know what valence electrons and valency of cesium are, aren’t you? But for this is not found in a free state in nature due to its high reactivity behavior so that it is abstracted from different compounds (mostly from salts).Īs we know how much cesium is being used in the world ofĬhemistry, so we must have very good proper information about its electronic properties to survive in the world of chemistry and that’s why you are here to Reactive alkali metals of group 1 with an atomic number 55 in the periodic table. We believe the article would prove to be beneficial in the study of same.Caesium or Cesium, a chemical element with a symbol Cs, is one of the highly So, this is all about the Caesium chemical element to understand the electron configuration and other properties of the element. So, Caesium has a huge role to play in energy production and conversion. This generator does the particular job of converting the heat energy to electricity. So, for the same reason, Caesium remains the highly demanded product in the oil industry.įurthermore, Caesium has other numbers of usages such as in the atomic clocks or in the production of thermionic generators. It works as the drilling fluid for the same oil extraction purpose. The major use of the Caesium goes to the oil industry where the chemical element is used in oil extraction related activities. Well, Caesium is a highly practical chemical element that has a number of significant applications. For instance, the electron configuration helps in figuring out all the other relevant chemical properties of Caesium or any other element. This configuration of the electron for the chemical element is useful in the further research of the chemical element. This electron distribution equation is what becomes the electron configuration of the element. The electron configuration of Caesium is nothing but the representation of the electrons of the element to its orbitals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
